Extensive internal and external feedback is leading the Food and Drug Administration to further refine a reorganization plan to include clear responsibilities for the leader of the Human Foods Program and significant changes for its Office of Regulatory Affairs.
When FDA unveiled plans for a new Human Foods Program in late January, many stakeholders criticized its lack of direct line authority for the newly-created deputy commissioner of foods and the continued siloing of offices in the agency. Many of those concerns are now being addressed through an updated flow chart of the proposed reorganization as FDA works toward finalizing the reorganization plan this fall.
FDA Commissioner Robert Califf told Agri-Pulse that while many people disagreed with specifics of the plan, the “overarching theme of almost everyone was clarity of decision-making power and purpose so they can just do their jobs.”
FDA Principal Deputy Commissioner Janet Woodcock told Agri-Pulse the agency has conducted more than 40 listening sessions in the past several months to hear reactions to the proposal from FDA's staff as well as their thoughts on external criticism of the proposed plan. FDA also put together an implementation and change management group, which currently has about 50 people involved, to help with designing the reorganization.
In the wake of last year's infant formula crisis, caused when a plant shutdown created supply shortages across the country, FDA came under fire for not having a person in charge of the human side of the foods program at the agency. The Reagan-Udall report called for clear leadership and revealed a history of disjointed organizational structure.
Woodcock, who is overseeing the reorganization efforts, said the proposed plan is “flattening the organization.” Instead of the previous hierarchy and its multiple steps or layers of different programs, different divisions and groups will work “hand-in-glove” with each other as defined by the organizational flow chart.
“People were really eager to have clear decisional lines of authority and to have clear priorities so we’re not just doing risk assessment. We’re going to do very formal risk prioritization, so people know what are the highest priorities, and they’re not trying to respond to multiple different priorities,” Woodcock said.
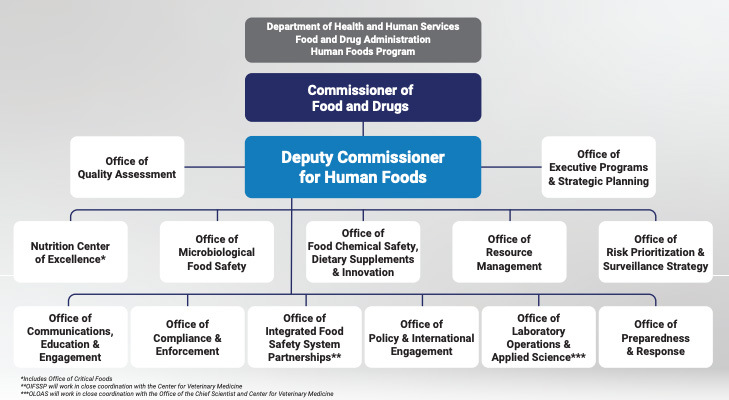
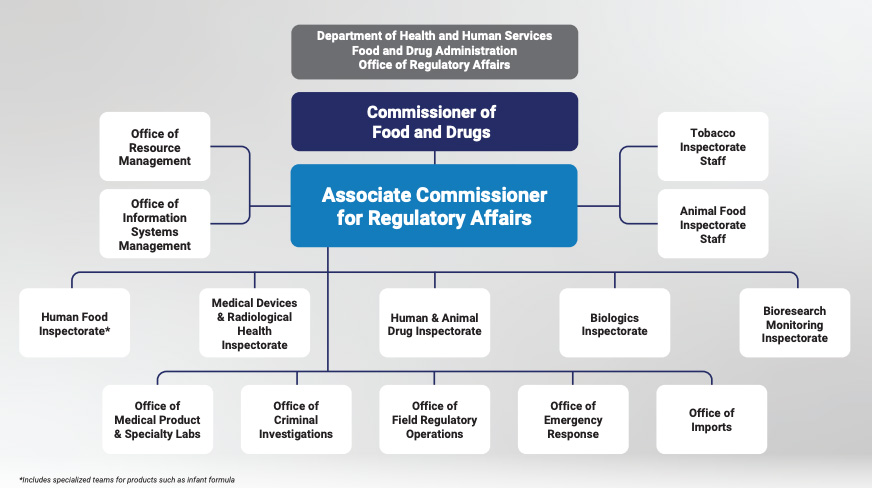
Califf said FDA is in the “final stages of interviews” for the deputy commissioner position. The agency also will name a new associate commissioner for regulatory affairs to lead ORA through the proposed changes.
“The head of the Human Foods Program will have accountability and authority over resources and priorities,” Califf said.
FDA’s newest proposal further defines the core mission of ORA as overseeing investigations, inspections and imports for all FDA-regulated products. The plan also transitions certain functions under the Office of Security and Emergency Management, currently in the Office of Operations, to ORA, FDA said. The agency also may rename the ORA to more accurately reflect what the division does, he said.
Woodcock said another major change developed through feedback on the plan was the need to reform how the laboratories are managed. “Certainly, all of us thought the laboratories was something that we didn't go in thinking that was as much of an issue;" however, the listening sessions revealed otherwise.
FDA now plans to realign the eight human and animal food laboratories currently managed by ORA into the Human Foods Program into four “super laboratories” at FDA’s current Center for Food Safety and Applied Nutrition (CFSAN) to form a unified food lab system.
FDA said, “The labs will report to a member of the executive leadership team under the deputy commissioner for human foods, who will work closely with the chief scientist and the Center for Veterinary Medicine (CVM) director to coordinate on research priorities. These labs will remain open and in the same geographic location under the proposal.”
Don’t miss a beat! It’s easy to sign up for a FREE month of Agri-Pulse news! For the latest on what’s happening in Washington, D.C. and around the country in agriculture, just click here.
Califf stressed the agency is trying to “create an organization that is set up to handle” future needs of the agency, based on a five- to 10-year outlook.
Many industry stakeholders welcomed the latest round of updates to FDA’s plan.
Joseph Scimeca, senior vice president of regulatory and scientific affairs for the International Dairy Foods Association, said in an email to Agri-Pulse that IDFA "agrees with the agency’s continued focus on breaking down silos, unifying its food program and emphasizing that the deputy commissioner for the human foods program will be responsible for coordinating closely with the FDA lead for the animal foods program and the Office of Regulatory Affairs.
"We appreciate the commissioner reiterating that the deputy commissioner will have full responsibility for setting the strategic direction of HFP, and for formulating and executing the program’s budget,” Scimeca said.
He also said IDFA welcomes the ORA changes, “particularly the refocusing of the office on inspections and outbreak investigations while also addressing duplication of efforts and overhead.”
Sciemca said IDFA is encouraging FDA to “reassign cooperative programs staff — particularly those that oversee the Grade A dairy program — to the Human Foods Program.”
Brian Ronholm, director of food policy at Consumer Reports, said in an email to Agri-Pulse that it appears FDA is “heading in the right direction on the reform ideas, especially related to the labs, the state and local partnership and functions compliance.”
FDA said it plans to unify state and local food safety partnerships and certain aspects of its international food safety partnerships into an Office of Integrated Food Safety System Partnerships at the new HFP.
Cathy Burns, CEO of the International Fresh Produce Association, said the “bold changes” announced by Califf “align with the reform IFPA has called for over the past year, individually and as part of broad stakeholder coalitions.”
“Along with the other changes announced, the unified laboratory enterprise will streamline sampling and testing issues faced by the fresh produce industry.”
She said the possible name change for ORA and the progress in recruiting for key leadership positions are “all signs that FDA is serious about rethinking how to best serve public health.”
For more news, visit www.Agri-Pulse.com.


